A 5th revision post for the British Beekeeping Association’s Module 3 Honey bee Pests, Diseases and Poisoning exam, which I plan to take in March.
B1. you suspect that a colony of honeybees has Nosema disease; describe fully
a) the signs and symptoms that aroused your suspicions
Nosema apis and Nosema ceranae are extremely common spore-producing fungus parasites, which are very likely to be affecting your bees. It’s also likely that you won’t have a clue they’re there, as the symptoms are not obvious.
Nosema spores enter via the mouth parts of a bee and travel into its mid-gut. In the case of an older foraging bee, they will typically pick up spores from trophallaxis (food-sharing) with other bees or drinking water sources contaminated by bee droppings. Younger house bees are more likely to be infected as they clear away excrement or clean comb in the hive. Normally bees are very clean creatures, but if long periods of bad weather prevent cleansing flights, a bee suffering from dysentery just can’t hold it in anymore. The younger house bees then try to clean up the mess, ingesting the destructive spores in the process.
Photo below of Nosema ceranae from http://www.biosecurity.govt.nz.

How nosema develops inside a bee
The spores invade her digestive epithelial cells, which line her mid gut – the function of the mid-gut is to produce digestive enzymes which enable the bee to digest the pollen and nectar in its diet. The spores germinate and feed happily away on the content of these cells, rapidly multiplying by cell division until the gut contains 30-50 million nosema spores once the infection is fully developed.
This impairs the digestion of pollen, shortening the lifespan of the bee (sources disagree on how much, with various books/websites quoting 50%, 10-50% and up to 78% – the earlier the bee picks up the spores, the more dramatically its lifespan is likely to be shortened). Infection is more devastating in younger bees, which will not be able to digest enough pollen to develop their hypopharyngeal glands and be able to produce brood food.
Nosema ceranae also suppresses the vitellogenin (Vg) gene in nurse bees, a gene which paces the onset of foraging and influences worker longevity. When Vg expression is suppressed, nurse bees transition to become foragers more quickly than a healthy bee would, resulting in a shorter lifespan.
Within around five days in each individual epithelial cell, spore reproduction ceases and the cell is ruptured and destroyed. Some of the spores invade new host cells, others pass through into the midgut and then down the small intestine into the rectum. From there they are excreted in the bee’s faeces and can contaminate other bees.
Diagram below showing nosema apis lifecycle from Scientificbeekeeping.com, © Springer Life Sciences. The spore injects its contents into a gut epithelial cell, multiplies, and eventually causes the cell to burst and release the new spores back into the gut. Nosema can also reproduce “vegetatively” cell to cell.

Just how bad is nosema?
How badly the colony is affected as a result will depend on the percentage of bees infected – if a high percentage are infected then it can have a serious effect on the colony. The symptoms to watch out for are a colony that fails to build up in spring, with lower brood production than you would normally expect. Honey production is also affected, due to a decrease in foraging time spent as bees die off early. Colonies usually survive and have fewer infected individuals once warmer late spring weather appears and bees are able to fly out to excrete away from the hive.
Some slight differences have been observed in the symptoms caused by the two species Nosema ceranae and Nosema apis. Dysentery spots and visible adult bee mortality in front of the hives are sometimes associated with N.apis but reported to be generally absent with N.ceranae. N.apis appears to thrive in colder weather whereas N.ceranae, which originated in Asia, seems to prefer warm climates and is more common during summer months.
Could nosema ceranae be implicated in colony collapse disorder (CCD)?
There is some evidence that N.ceranae infections are more voracious than Nosema apis; in Spain N. ceranae infections have been observed to be characterised by a progressive reduction in the number of bees in a colony until the point of collapse. As the colony becomes under increasing pressure, secondary diseases frequently appear, including chalk brood and American foul brood.
There has even been some speculation that N. ceranae could be a contributing factor in CCD – if the colony breaks down, the queen can be found surrounded by a few bees, confusedly attending to brood that is already sealed. Infected bees tend to altruistically prevent spreading the infection by flying away and not returning to the hive. Newly emerged bees may not test positive for nosema, allowing the culprit to go undiscovered. However, more recent research has failed to confirm a link and researchers in the US have concluded that it is not the primary cause of CCD.
b) how you would confirm that the disease is present
Definite confirmation requires microscopic examination of the abdomens of older adult bees, which can be collected from the hive entrance. Infection is highly variable in a colony, but foragers tend to contain the higher spore loads. Collecting bees leaving to go on cleansing flights rather than returning will be more effective. Send a sample of 30 bees to a microscopist or the UK National Bee Unit service in a small sturdy box (not plastic, which causes rapid sample degradation).
From a Beecraft magazine article – Bee Craft, Nosema ceranae Jan 2008:
“Abdomens of worker bees are ground up in a mortar with a little water and a drop of the resulting liquid spread onto a microscope slide. The tell-tale rice grain shapes can be seen using a microscope with a magnification of 400x. If you take a large enough sample of bees, you could probably detect minute levels of Nosema in many colonies. For a realistic result, it is recommended that you use around 30 bees.”
Diagram below from Scientificbeekeeping.com. The difference between the two species is very slight, but N.apis tends to have a more transparent centre and N.ceranae more of a slender, ‘bent’ shape.

The extent of infection is determined by counting the spores on a microscope grid and calculating the average number of spores per area and estimating from that the number of spores per bee.
Under the light microscope the spores of N. apis and N. ceranae appear as white/green, rice shaped bodies. Telling the difference between the two species is not easy, but some say an expert eye can spot it. On average N.apis spores are one micron larger in length than N.ceranae. Additionally N.ceranae spores can appear ‘bent’ and thinner than N.apis spores; however treatment is the same for both species. Genetic examination is the only reliable technique to distinguish between the two.
c) the action that should be taken to treat the disease if confirmed
Fumadil B, the antibiotic previously used for treating nosema, has recently become no longer approved for sale in the UK (since December 2011). Without this treatment at our disposal, moving the colony onto fresh comb that contains no nosema spores is the best option.
Our best defence against nosema is good husbandry practices such as regularly changing brood comb, avoiding crushing bees through clumsy handling (this can release millions of nosema spores inside the hive) and maintaining strong well fed colonies, headed by young prolific queens.
As always, avoid moving brood frames between hives and do not feed honey from other colonies to bees – nosema spores can remain latent over a year on combs and for three to four months in honey. Wash hive tools between hives with a solution of one part washing soda to five parts water (e.g. 1kg of soda dissolved in 5 litres of water). Bee keepers should also consider re-queening susceptible colonies with queens from more tolerant stocks of bees better able to cope with Nosema infection.
d) how you would get the bees onto new sterile combs to allow you to remove the original frames for sterilisation – give two methods.
Bailey comb exchange – all infected brood comb is removed gradually, over a period of about a month. A new brood box filled with foundation is placed over the existing one. Once the bees have drawn out some of the foundation, the queen is found and placed in the new brood box. A queen excluder is placed between the two brood boxes, so that she cannot move down to the old one. Heavy 2:1 mixture syrup should be fed to encourage the workers to draw out new comb quickly.
Three weeks later, once the larvae in the old brood combs have emerged, these frames can be removed and either burned or fumigated with acetic acid. Acetic acid is very corrosive stuff, so beginners should be cautious when trying this and seek advice from more experienced beekeepers first. The Bailey comb exchange is best done in early spring, such as March in the UK.
See the FERA Beebase website’s Replacing Comb factsheet for a more detailed explanation of the Bailey exchange method.
Shook-swarm – this is a dramatic spring clean for the bees, removing all their old brood comb with larvae cocoons/faeces, varroa and possibly nosema spores and giving them fresh new foundation to build on. Best done on a warm, sunny day in early Spring.
First move your hive to one side and put a ‘new’ hive and foundation frames in the location of your old one, with a queen excluder on top of the floor (between the floor and the brood box). The queen excluder stops the queen absconding in the first week, and putting the new hive in the old location ensures that any foragers return to the right place.
Next, find the queen (easy, right?!) and put her safely in your pocket in a queen cage. Remove four frames from the centre of the new hive and place them to one side. Shake all the workers into the gap and return the queen. The old brood comb can now be burnt. Feed 2:1 strength sugar syrup to the colony to help them draw out new comb quickly and return regularly to top this up. The queen excluder can be removed a week later.
See the FERA Beebase website for a free downloadable shook-swarm factsheet which is worth following step-by-step if you’re shook-swarming for the first time.
B2. a) give a simple account of the structure and function of the alimentary, excretory and respiratory systems of the adult honeybee worker
- Ailmentary systems
Digestion and excretion are the functions of the alimentary canal and its associated glands. Immediately inside the bee’s mouth the canal extends into a cavity with muscular attachments to the front of the head, so that it can expand and contract, providing small amounts of suction to help pass food from the proboscis and into the oesophagus. Waves of contractions are produced by muscles in the oesophagus, working nectar back into the bee’s initial ‘honey stomach’ or ‘crop’, where it is stored for a while.
At the end of the honey stomach is an important valve called the proventriculus, which stops the nectar going further until it’s needed.The proventriculus has four lips that are in constant movement, sieving out solids such as pollen grains and spores from the nectar and passing them back as a fairly dry lump into the ventriculus (mid-gut).
When the bee needs sugar, the proventriculus opens up and allows some nectar through into the ventriculus, where enzymes break the nectar down into molecules small enough to be passed through the gut wall into the haemolymph.Any waste residue is passed into the small intestine and held as faeces in the rectum until the bee is able to leave the hive for a poop. During long periods of cold weather in winter the rectum can expand to almost the whole length of the abdomen before the bee is able to fly out. This is when bees are more likely to have trouble ‘holding it in’, especially if they are suffering from dysentery.
Illustration below by R.E. Snodgrass from http://www.extension.org. Alimentary canal of worker (Phy-Rect), together with pharyngeal glands (1Gl), and salivary glands of head (2Gl) and of thorax (3Gl), as seen by cutting body open from above and pulling the ventriculus (Vent) out to left.

- Excretory systems
As a bee digests its food, the food breaks down and the resulting products are circulated by the bee’s haemolymph (a bee’s equivalent to our blood). The useful products provide energy and bodybuilding substances. The waste products need to be expelled by the excretory system, which acts as a sophisticated filtering system. As well as removing waste substances, it also adjusts the levels of particular substances in the haemolymph, ensuring the correct balance between water and salts and that osmotic pressure and acidity remain within narrow limits.
- Respiratory systems
The bee breathes through tubes called trachea, which carry oxygen throughout the bee’s body to where it’s required. In more complex animals such as mammals, oxygen is transported to body tissues by blood, but the haemolymph of insects is not involved in transporting oxygen.
The trachea open to the air around the bee through holes in the cuticle called spiracles, which in many cases are provided with a closing mechanism. Air enters the tracheal system through the spiracles and is diffused around the bee’s body.
Diagram below of the internal anatomy of a honey bee found at chdphd.com/PhD/Chapter2.php, adapted from Dade (1977) and Winston (1987).

b) explain how Acarine, Nosema and Amoeba affect these systems.
- Acarine is a small mite named Acarapis woodi which lives in the main thoracic trachea (respiratory tubes) of the honey bee. A fertilised female will find her way into a bee’s trachea and lay 5-7 eggs soon after the bee emerges from its cell – she prefers her host to be only one to two days old. There is some evidence that she prefers drones as hosts, due to their larger tracheas.
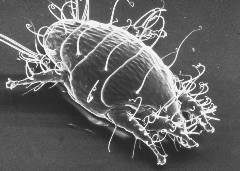
Tracheal mite/tiny guinea pig. Courtesy The Food and Environment Research Agency (Fera), Crown Copyright.
The eggs hatch in about five days, and the little larvae (which Ted Hooper describes in Guide to Bees and Honey as reminding him of “tiny guinea pigs”) develop into adult mites about nine days later. The mite family feed on the haemolymph (blood equivalent) of the infested bee through the walls of the trachea, piercing it with their mouth parts. Their life cycle is complete with fourteen to nineteen days, after which they mate in the trachea and the fertilised females leave to start a new family in other young bees.
Grabbing a new host
The female mites find a new host by leaving the trachea, clinging onto one of the bee’s hairs with one or two hind legs, and then waving their remaining legs around until a suitable young bee less than nine days old comes along. She will be able to tell by the smell of the bee and also because the trachea is protected by hairs which are not so dense in young bees. She grabs the hair of her new host with her front legs and is drawn into the first spiracle through wing vibrations and puffs of air. Guinea pigs never did anything so dastardly!
Effect on the colony
The poor bee can have a trachea stuffed full of the mites, which has the effect of blocking its trachea, shortening its lifespan. Some honey bee sub-species are more susceptible than others – acarine is a bigger problem in the US, where many beekeepers use Italian/New Zealand crosses. Bees native to the UK appear to have evolved a tolerance to the mite.
It is thought that acarine usually weakens a colony and makes it susceptible to other diseases rather than killing colonies outright. The National Bee Unit give a guideline figure for the U. K. that if 30% of bees in a colony are infested then the colony will die in the following spring, but say that generally in Europe a realistic threshold could be set at about 10%.
It was probably a viral infection such as Chronic Bee Paralysis Virus and not acarine (as was originally thought) that killed so many colonies in the infamous Isle of Wight disease outbreak of 1904.
Diagram below of tracheal mite life cycle found on the Mid Atlantic Apiculture Research Consortium website, @MAARECnews

- As described above, Nosema spores multiply in the ventriculus (reaching 30 – 50 million spores) and impair the digestion of pollen, thereby shortening the life of the bee.
The spores germinate in an ingenious way – inside each spore is a coiled filament that is attached to one end of the spore. This filament everts quickly from the spore and injects a host mid-gut cell, pouring the contents of the spore (nucleus, membranes, etc.) into the host cell cytoplasm.
- Amoeba is caused by a parasite named Malpighamoeba mellificae. Its cysts are ingested by nurse/cleaning bees and germinate in the rectum.
The spores migrate to the malpighian tubules (the bee’s ‘kidneys’), attack the cells lining the tubules and after 3-4 weeks will divide to create more cysts, that then accumulate in the rectum and are excreted. The cycle then begins again when hive bees ingest the cysts whilst cleaning.
So the cysts take longer to develop than nosema spores – and the number produced is also sixty times less. A bee also has numerous malpighian tubules, so not all the tubules are likely to be damaged.
Whether amoeba infection has much effect on the bee is not clearly known. However, infections are very often found in association with nosema and it is likely that a dual infection will be more damaging to the health of the honey bee. Considering how many really nasty parasites and pests honey bees have to fight, amoeba cysts appear to be pretty benign parasites. Hurray! Their cuddly round shapes are often seen under the microscope when examining a sample for nosema spores.
Microscope picture below from Scientificbeekeeping.com. Amoeba cysts are larger and more oval than nosema spores, and don’t glow in the center.

References:
- Bee Craft article, Nosema ceranae, Jan 2008
- Guide to Bees and Honey, Ted Hooper & Margaret Thomas, Northern Bee Books (2010)
- The Honey Bee Around & About, Celia F Davis, Bee Craft Ltd, (2009)
- Keeping Healthy Honey Bees, David Aston & Sally Bucknall, Northern Bee Books (2010)
- Mid Atlantic Apiculture Research Consortium (MAAREC), Bee diseases and their control (2005)
- Mid Bucks Beekeepers Association, Module 3 exam notes
- National Bee Unit Beebase website
- ‘Nosema ceranae, a Causative Agent of Nosemosis in the UK‘, BBKA News No:216 – August 2013, p9-10
- Practical Beekeeping, Clive de Bruyn, Crowood Press (1997)
- Scientificbeekeeping.com, Randy Oliver

Please come to Canada and be my beekeeper!
LikeLike
I wish!
LikeLike
Excellent, text book account.
LikeLike
Thanks – but that’d be because I read all the textbooks I could find!
LikeLike
Wow, that’s a lot to learn…glad I’m not going to be tested on it. I’m just trying to get through the first winter with my bees. Dec. 9, they were flying well, still bringing in pollen.
LikeLike
It is a lot – and some people even take two or three different exams on the same day! No way I could attempt that. Good to hear yours are doing well, wonder if it’s still the ivy pollen keeping them going or something else now.
LikeLike
The ivy is still blooming, At the rate the bees are bringing in pollen, I would expect to see dozens of bees on the vines…could be they are not using the vines convenient for me to see. We’re in a rural setting with different types of brush and wild stuff growing.
Have you ever seen bees getting propolis? I saw some sap dripping out of a fir tree today but no bees around it. I’d LOVE to get a video of that.
I’m starting my beginning Master Beekeepers program at the first of the year…until they kick me out for being anti-established methods. We’ll see how it goes. 🙂
LikeLike
Your climate is obviously warmer than ours, the ivy is over here and frosts have set in. I’ve very occasionally seen foragers returning with propolis, but never been lucky enough to spot them collect it! Apparently it’s nibbled off by other bees once they return to the hive and used immediately to seal up gaps or varnish cells etc.
Enjoy your Master Beekeeping program! Let us know how it goes 🙂
LikeLike
Fascinating – especially the bee anatomy diagram. RH
LikeLike
It is a great diagram. If I take the British Beekeeping Association’s Biology exam I’m going to have to learn how to draw one of those from memory!
LikeLike
Pingback: How Honeybees Fly – muscles, wings, and stomach crunches | Brookfield Farm Bees & Honey Blog